Links to external sources may no longer work as intended. The content may not represent the latest thinking in this area or the Society’s current position on the topic.
Macropinocytosis in physiology, disease and therapy
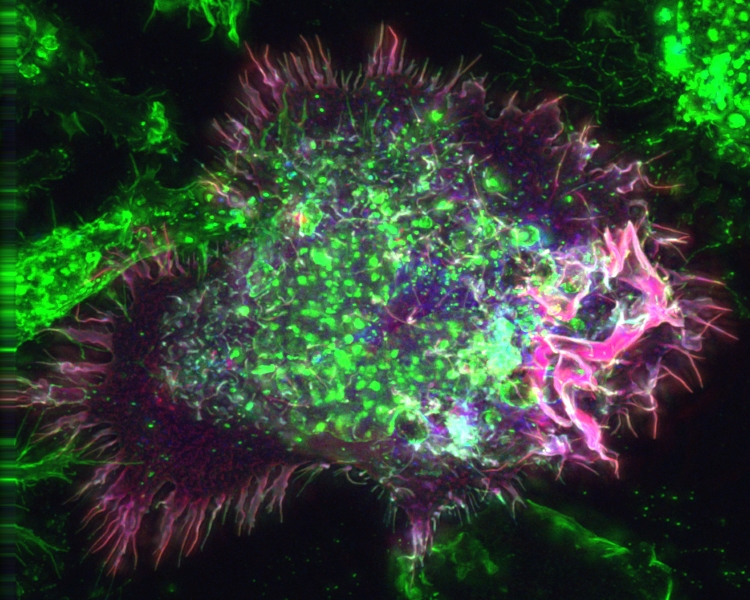
Theo Murphy international scientific meeting organised by Dr Jason King and Professor Joel Swanson.
The engulfment of extracellular solutes or particles by macropinocytosis is an important process with newly appreciated significance for fundamental cell biology, cancer, infectious diseases and therapeutics. This first-of-its-kind meeting devoted to macropinocytosis will bring together experts from disparate fields with a shared interest in the biology of macropinosome formation and trafficking, with the goal of fostering collaboration and building a community.
Speaker abstracts and biographies can be found below. Recorded audio of the presentations is also available below. An accompanying journal issue for this meeting was published in Philosophical Transactions of the Royal Society B.
Attending this event
This meeting has taken place.
Enquiries: contact the Scientific Programmes team
Organisers
Schedule
Chair

Professor Colin Watts FMedSci FRS, University of Dundee, UK

Professor Colin Watts FMedSci FRS, University of Dundee, UK
Colin Watts trained at the Universities of Bristol and Sussex, UK and then at the University of California, Los Angeles and the MRC Laboratory of Molecular Biology, Cambridge before starting his own lab at the University of Dundee in 1986 where he is currently Professor Emeritus in the School of Life Sciences. His lab has worked mainly on antigen uptake, processing and presentation in the immune system and sporadically on macropinocytosis, showing for example that it could be selectively inhibited by amilorides and triggered in dendritic cells by pathogen derived signals.
| 09:05 - 09:30 |
Macropinocytosis and cellular growth control
Macropinocytosis has long been implicated as a mechanism for growing cells to assimilate extracellular nutrients. Studies of murine macrophages and embryonic fibroblasts showed that activation of the growth-associated metabolic regulator mechanistic target of rapamycin complex-1 (mTORC1) in response to growth factors and extracellular amino acids requires internalization of amino acids by macropinocytosis. This led the group to propose that macropinocytosis is necessary for mTORC1-dependent growth of metazoan cells, as both a route for nutrient delivery into lysosomes and a platform for growth factor-dependent signalling to mTORC1 via PI 3-kinase (PI3K) and Akt. A functional actin cytoskeleton is required for macropinocytic cup formation and activation of mTORC1 by growth factors and amino acids. Although activation of Akt by some growth factors occurs independent of the actin cytoskeleton, maximal activation of Akt by stimuli that elicit weak Akt responses requires that cells be capable of forming macropinocytic cups or circular dorsal ruffles. This indicates that growth factors and chemokines which trigger low maximal levels of Akt activity require actin-based macropinocytic cup formation for localized amplification of PI3K-dependent responses. In this way, ruffles and cups could organize signalling by extracellular ligands into stochastic, structure-dependent cascades of chemical reactions that stimulate cell growth or orient cell migration. 
Professor Joel Swanson, University of Michigan, USA

Professor Joel Swanson, University of Michigan, USAJoel Swanson, Professor of Microbiology and Immunology, trained at Rutgers University (BA, Biology), Ohio State University (M.S., Botany), and Princeton University (PhD, Biology), followed by post-doctoral studies at Rockefeller University and Columbia College of Physicians and Surgeons. He served on the faculty at Harvard Medical School and the University of Michigan Medical School. His laboratory has a longstanding interest of heterophagy, which is the cellular ingestion of extracellular particles and solutes by macropinocytosis, phagocytosis and endocytosis, and the subsequent degradation of ingested materials in lysosomes. His approach emphasizes molecular and microscopic studies of the spatial organization of cytoplasm in cells. Recent work has demonstrated roles for macropinocytosis in the regulation of cell growth in macrophages and other cell types. |
|
|---|---|---|
| 09:30 - 09:45 | Discussion | |
| 09:45 - 10:15 |
Constitutive vs inducible macropinocytosis in macrophages
Like other cells, macrophages respond to growth promoters by ruffling their membrane, which in turn promotes macropinocytosis. However, the surveillance role of macrophages requires ongoing sampling of their environment, which is performed by constitutive macropinocytosis. The latter occurs continuously, even in the presence of growth promoters, generating smaller macropinosomes. Constitutive macropinocytosis requires extracellular calcium and is mediated by calcium-sensing receptors. Both inducible and constitutive macropinocytosis depend on actin and on phosphatidylinositol 3,4,5-trisphosphate (PIP3), but only the inducible form is sensitive to amiloride analogues. Constitutive macropinocytosis is active in anti-inflammatory macrophages, but negligible in pro-inflammatory macrophages, such as those polarized by treatment with GM-CSF, LPS and/or IFNγ. Inflammatory macrophages have reduced levels of PIP3, which accounts at least in part for their inability to perform constitutive macropinocytosis. Oxidized LDL (oxLDL), the source of the vascular plaque responsible for heart attacks and strokes, is taken up by macrophages via scavenger receptors. Unlike conventional receptor-mediated endocytosis, oxLDL uptake requires actin polymerization. The evidence indicates that constitutive macropinocytosis is largely responsible for oxLDL internalization by non-inflammatory macrophages, in a scavenger receptor-dependent manner. The group has termed this process 'receptor-assisted macropinocytosis'. 
Professor Sergio Grinstein, The Hospital for Sick Children, Canada

Professor Sergio Grinstein, The Hospital for Sick Children, CanadaDr Sergio Grinstein completed his PhD in 1976 at the Centro de Investigacion y Estudios Avanzados, in Mexico City. He then spent two years as a post-doctoral fellow at the Hospital For Sick Children in Toronto, followed by a year in the Department of Biochemistry at the Federal Institute of Technology in Zurich. He is currently working at the Hospital For Sick Children in Toronto and has been Professor of Biochemistry at the University of Toronto since 1988. Dr Grinstein is interested in the cell physiology and biophysics of innate immunity, particularly phagocytosis and host-pathogen interactions. |
|
| 10:15 - 10:30 | Discussion | |
| 10:30 - 11:00 | Coffee | |
| 11:00 - 11:30 |
Macropinosome formation, maturation and membrane recycling
Macropinocytosis is an unusual form of endocytosis that has fascinated cell biologists for decades. Recent advances in live cell imaging have allowed the study of this process in more detail. Professor Donaldson's lab has focused on how plasma membrane is shaped to form the macropinosome, how the macropinosome is brought into the cell interior and how membrane is sorted out from the maturing macropinosome. They find a requirement for microtubules and dynein for macropinocytosis that is driven by Ras. In addition, Arf6 and its effector, the JIP3 microtubule motor scaffold protein are also involved in macropinosome formation and transport through the lamellar region. Once past the lamellar actin/myosin arc, actin is shed from the macropinosome and it begins to undergo cargo sorting for membrane recycling back to the cell surface. Cargo entering into the macropinosome are mostly clathrin-independent cargo proteins and during sorting, select proteins (CD98 and CD147) leave the macropinosome for recycling. Retromer components are recruited onto the macropinosome during this sorting out of cargo. Similar sorting of cargo is observed in other cells but the large size of the macropinosome enables us to track cargo sorting in space and time. 
Professor Julie Donaldson, National Institutes of Health, USA

Professor Julie Donaldson, National Institutes of Health, USAJulie Donaldson received her PhD from the University of Maryland in 1988 and did her postdoctoral research training at NIH before starting her own lab in 1995 at the National Heart, Lung and Blood Institute at the NIH. She is currently a Senior Investigator in the Cell Biology and Physiology Center. Her research program focuses on understanding the mechanism and physiological function of clathrin-independent endocytosis (CIE) and the subsequent routing and fate of endocytosed membrane proteins. CIE is the entry mechanism for many cell surface proteins including cell adhesion molecules, ion channels, nutrient transporters and microbial pathogens. Macropinocytosis is another form of CIE that provides a unique platform for following membrane dynamics. |
|
| 11:30 - 11:45 | Discussion | |
| 11:45 - 12:15 |
Learning how to form and process macropinosomes with Dictyostelium amoeba
Macropinocytosis requires the co-ordinated regulation of both the cytoskeleton to form the cup-shaped protrusions, as well as the vesicular trafficking machinery to process them after internalisation. The group is trying to understand both these processes using Dictyostelium amoeba, which use macropinocytosis for feeding. Macropinocytic cups are able to self-assemble stochastically, without any external physical scaffolds or localised signals. This requires cells to generate a ring of protrusion, driven by localised actin polymerisation, that encircles a static membrane domain that defines the cup interior. Dr King will discuss a new mechanism by which the Ras and Rac small GTPases differentially regulated across the protrusive rim and cup interior, in order to spatially modulate the cytoskeleton and generate protrusions that efficiently engulf fluid. After engulfment cells must rapidly recycle any surface components before they are degraded, whilst orchestrating a complex series of trafficking steps that ensure the efficient processing of the internalised material. Dr King will therefore also discuss how the group is using the amoeba model to dissect these processes to provide a mechanistic understanding of macropinosome maturation. 
Dr Jason King, University of Sheffield, UK

Dr Jason King, University of Sheffield, UKJason King is currently a Royal Society University Research Fellow at the University of Sheffield, having moved there in 2013 to start his independent group. Their main interests are understanding how cells both form, and regulate the maturation of macropinosomes and phagosomes. Prior to this Dr King worked as a postdoc in the laboratory of Robert Insall at the Beatson Institute for Cancer Research in Glasgow, UK, studying the cytoskeleton and cell migration, before developing interests in autophagy and trafficking. This followed on from his PhD work in the lab of Adrian Harwood at Cardiff University, studying how lithium treatment (still widely used to tread bipolar disorder) affects cellular signalling and in particular phosphinositide signalling. The group’s current work on macropinocytosis brings all these interests together in order to determine how both the cytoskeleton and vesicular trafficking are coordinated to facilitate fluid uptake and processing. |
|
| 12:15 - 12:30 | Discussion |
Chair

Professor Sergio Grinstein, The Hospital for Sick Children, Canada

Professor Sergio Grinstein, The Hospital for Sick Children, Canada
Dr Sergio Grinstein completed his PhD in 1976 at the Centro de Investigacion y Estudios Avanzados, in Mexico City. He then spent two years as a post-doctoral fellow at the Hospital For Sick Children in Toronto, followed by a year in the Department of Biochemistry at the Federal Institute of Technology in Zurich. He is currently working at the Hospital For Sick Children in Toronto and has been Professor of Biochemistry at the University of Toronto since 1988.
Dr Grinstein is interested in the cell physiology and biophysics of innate immunity, particularly phagocytosis and host-pathogen interactions.
| 13:30 - 14:00 |
Unexpected roles for macropinocytosis in cell migration
The migration of immune cells is guided by specific chemical signals, such as chemokine gradients. Nevertheless, their trajectories can also be diverted by physical cues and obstacles imposed by the cellular environment, such as topography, rigidity, adhesion, or hydraulic resistance. On the example of hydraulic resistance, it was shown that neutrophil preferentially follow paths of least resistance, a phenomenon referred to as barotaxis. The group here combined quantitative imaging and physical modelling to show that barotaxis results from a force imbalance at the scale of the cell, which is amplified by the acto-myosin intrinsic polarization capacity. Strikingly, macropinocytosis specifically confers to immature dendritic cells a unique capacity to overcome this physical bias, which enhances their space exploration capacity in vivo and promotes their tissue-patrolling function. Conversely, mature dendritic cells, which down-regulate macropinocytosis, are sensitive to hydraulic resistance. Theoretical modelling predicts that being barotactic helps them avoid dead-ends while migrating to lymph nodes to initiate the adaptive immune response. The group concludes that the physical properties of the microenvironment of moving cells can introduce biases in their migratory behaviours but that specific active mechanisms such as macropinocytosis have emerged to diminish the influence of these biases, allowing motile cells to reach their final destination and efficiently fulfill their functions. 
Professor Ana-Maria Lennon-Duménil, Institut Curie, France

Professor Ana-Maria Lennon-Duménil, Institut Curie, FranceAna-Maria Lennon-Duménil is a cell biologist, team-leader in the ‘Immunity and Cancer’ Department at the Curie Institute in Paris since 2004. Her career has focused on combining cell biology with other disciplines such as genetics (PhD), biochemistry and organic chemistry (post-doctoral stage), and biophysics (group-leader) to address key questions concerning immune cells. She has successfully coordinated for eight years a consortium named ‘Dendritic Cell Migration in Confined Environments’ that includes Matthieu Piel (Institut Curie) and Raphaël Voituriez (UPMC), and generated more than 20 publications, some in very prestigious journals such as Cell, Science, Nature Cell Biol., Nature Cell Physics, Nature Comm. Her work as an independent investigator has opened a yet unexplored research line on the coordination of cell function and cell migration. |
|
|---|---|---|
| 14:00 - 14:15 | Discussion | |
| 14:15 - 14:45 |
Macropinocytosis: Ras and PIP3 driven endocytosis
Macropinocytosis probably originated as a feeding mechanism in unicellular organisms and is used in this way by amoebae today. Dictyostelium amoebae form several large macropinosomes per minute in rich medium and can take up a significant fraction of their entire volume of fluid per hour, with macropinocytosis accounting for more than 90% of this. Macropinocytic cups are organized around intense patches of activated Ras and PIP3 in the plasma membrane, whose size is regulated by the RasGAP, NF1. These patches recruit the SCAR/WAVE complex to their periphery, thus initiating a hollow ring of actin polymerization to form the walls of the macropinocytic cup. PIP3 is essential for efficient macropinocytosis in Dictyostelium, as in mammals, and we find that the downstream protein kinase, PKB, which is recruited to the plasma membrane by PIP3, is similarly important. The group has identified a number of targets for PKB, among which regulators of small G-proteins are prominent. Professor Kay will also describe how active-Ras/PIP3 patches are unexpectedly sensitive to disruption of cytoskeletal organization. 
Professor Rob Kay, MRC Laboratory for Molecular Biology, UK

Professor Rob Kay, MRC Laboratory for Molecular Biology, UKRob Kay’s group is interested in finding fundamental mechanisms of macropinocytosis using Dictyostelium amoebae and mammalian cells as models. Rob is a group leader at the MRC Laboratory of Molecular Biology in Cambridge. He studied Biochemistry at UCL and was an EMBO fellow in Freiburg before working at the Mill Hill laboratory of the Imperial Cancer Research Fund with John Cairns and Julian Gross. He is a member of EMBO and former Howard Hughes International Fellow. |
|
| 14:45 - 15:00 | Discussion | |
| 15:00 - 15:30 | Tea | |
| 15:30 - 16:00 |
Macropinosomes as launch-pads for inflammation in macrophages
Enhanced ruffling and macropinocytosis on pathogen-activated macrophages help to drive innate immune and inflammatory responses. These membrane domains function in tissue surveillance, antigen capture, receptor activation and as signaling domains for pro-or anti-inflammatory transcriptional programs. With the high temporal and spatial resolution of lattice light sheet microscopy for live cell imaging the group has been able to resolve new features of dorsal ruffles and redefine their dynamic closure and transition to macropinosomes. These features depict how different sized ruffles and macropinosomes can be made. Moreover, the group has defined roles for sequential Rab GTPases in the formation of LPS-induced, enlarged ruffles and macropinosomes. Toll-like receptor (TLR) signalling also occurs on early macropinosomes where the group has shown a TLR4/coreceptor/GEF/Rab complex recruits PI3K for Akt/mTORC1 signalling. This signalling pathway biases cytokine outputs and is a key mechanism for driving an M2-like macrophage program for resolution of TLR-induced inflammation. The highly dynamic, rapidly transitioning domains on early macropinosomes support TLR signalling and protein sorting that determines the outcome of inflammation. 
Professor Jenny Stow, University of Queensland, Australia

Professor Jenny Stow, University of Queensland, AustraliaProfessor Jennifer Stow heads the Protein Trafficking and Inflammation research group at The University of Queensland’s Institute for Molecular Bioscience (IMB) in Brisbane, Australia. Her tenure at IMB has included appointments as IMB Deputy Director (Research), Head of the Division of Molecular Cell Biology, NHMRC Principal Research Fellow and Wellcome Trust Senior Medical Research Fellow. Earlier appointments include as Assistant Professor at Massachusetts General Hospital/Harvard Medical School and postdoctoral fellow in the department of Cell Biology, Yale University School of Medicine, following award of her PhD from Monash University, Australia. The Stow group use multiscale microscopy and live cell imaging, coupled with biochemistry, to define key macrophage molecules, receptor signalling and trafficking pathways that regulate inflammation. |
|
| 16:00 - 16:15 | Discussion | |
| 16:15 - 18:30 | Poster session |
Chair

Professor Ana-Maria Lennon-Duménil, Institut Curie, France

Professor Ana-Maria Lennon-Duménil, Institut Curie, France
Ana-Maria Lennon-Duménil is a cell biologist, team-leader in the ‘Immunity and Cancer’ Department at the Curie Institute in Paris since 2004. Her career has focused on combining cell biology with other disciplines such as genetics (PhD), biochemistry and organic chemistry (post-doctoral stage), and biophysics (group-leader) to address key questions concerning immune cells. She has successfully coordinated for eight years a consortium named ‘Dendritic Cell Migration in Confined Environments’ that includes Matthieu Piel (Institut Curie) and Raphaël Voituriez (UPMC), and generated more than 20 publications, some in very prestigious journals such as Cell, Science, Nature Cell Biol., Nature Cell Physics, Nature Comm. Her work as an independent investigator has opened a yet unexplored research line on the coordination of cell function and cell migration.
| 09:00 - 09:30 |
How mammalian cells regulate the use of proteins as nutrients
The cells of mammalian organisms are surrounded by diverse nutrients, including low-molecular weight nutrients such as glucose and amino acids as well as various macromolecules. Cells can import free amino acids through cell surface transporters or recover amino acids through macropinocytosis and lysosomal degradation of extracellular proteins. While it was assumed that mammalian cells rely on uptake of free amino acids, the group demonstrates that macropinocytosis and lysosomal catabolism of extracellular proteins can support cell survival and proliferation in amino acid-deprived environments. Nutrient uptake is regulated by growth factors and their effectors, the Ras and PI3-kinase signalling pathways. Consistently, oncogenic mutations in Ras or PI3-kinase, which promote macropinocytosis, allow cancer cells to proliferate by exploiting extracellular proteins as nutrients. However, concerted inputs from growth factor signalling and protein catabolism also activate the nutrient-sensing kinase mTORC1, which blocks lysosomal degradation of ingested protein. Inhibiting mTORC1 results in increased catabolism of macropinocytosed proteins and enhances cell proliferation during nutrient-depleted conditions in vitro and within vascularly compromised tumours in vivo. This suggests that in response to Ras and PI3-kinase signalling, macropinocytosis dominates as a nutrient acquisition strategy when a decline in intracellular amino acid levels leads to inactivation of mTORC1. Together, these findings identify signalling pathways that regulate the usage of macropinocytosis of proteins as an amino acid source. 
Dr Wilhelm Palm, German Cancer Research Center, Germany

Dr Wilhelm Palm, German Cancer Research Center, GermanyWilhelm Palm leads the cell signalling and metabolism group at the German Cancer Research Center in Heidelberg. Wilhelm’s PhD research with Suzanne Eaton at the Max Planck Institute of Molecular Cell Biology and Genetics investigated roles of Drosophila lipoproteins in lipid metabolism and hedgehog signalling. During his postdoc with Craig Thompson at Memorial Sloan Kettering Cancer Center, he studied how growth factor signalling and the nutrient sensor mTORC1 regulate macropinocytosis of extracellular proteins as an amino acid source for mammalian cells. In his independent group, Wilhelm continues to study the reciprocal relationship of signalling and metabolism, with main focus on the regulation of endolysosomal nutrient acquisition pathways. |
|
|---|---|---|
| 09:30 - 09:45 | Discussion | |
| 09:45 - 10:35 | Short talks | |
| 10:35 - 11:00 | Coffee | |
| 11:00 - 11:30 |
Macropinocytosis through the lens of Ebolavirus
Viruses offer advantages over conventional cell biology probes being discreet particles made of a few well defined proteins. They are easily labelled with fluorescent tags and are distinct by electron microscopy. Furthermore, once inside the cell, virus gene expression is readily detected by antibody staining or PCR. Like other viruses, Ebola virus is an obligate parasite, relying on host proteins for infection, from the point of cell entry, through virus replication and finally egress from the cell. The group is focused on identifying host factors that control early steps of Ebola virus entry into cells. Earlier, the group was the first to demonstrate that Ebola virus is internalized into cells by a macropinocytosis-like mechanism. More recently, the group has developed high throughput screening systems, based on virus infection, to identify small molecules and siRNA-suppressed host factors that inhibit virus infection by blocking macropinocytosis. Using this approach the group has identified a novel class of small molecules that appear to potently and specifically inhibit Ebola virus infection by interfering with macropinocytosis while not affecting other endocytic pathways. The group has also identified host proteins involved in the autophagy pathway that have prominent roles early in virus entry that are also important in early macropinosome formation. Professor Davey will discuss the latest understanding of Ebola virus uptake into cells and compare and contrast virus uptake to general macropinocytosis mechanism. 
Professor Robert Davey, Texas Biomedical Research Institute, USA

Professor Robert Davey, Texas Biomedical Research Institute, USARobert A Davey, PhD is the Ewing and Halsell Scholar in the Department of Virology and Immunology at Texas Biomedical Research Institute in San Antonio, Texas. The institute is host to one of six active BSL4 (P4) laboratories in the USA. He came to San Antonio after a postdoc at the Harvard Medical School and then as an Assistant and Associate professor at the University of Texas Medical Branch in Galveston where he developed a strong interest in how viruses invade cells to establish infection. His present work is focused on the mechanism of Ebolavirus entry into cells, which occurs predominantly through a form of macropinocytosis. Using this virus as a probe, the group are revealing new aspects of macropinocytosis mechanism, identifying cell factors responsible for endocytosis and finding new small molecule inhibitors of this important process. |
|
| 11:30 - 11:45 | Discussion | |
| 11:45 - 12:35 | Short talks |
Chair

Professor Julie Donaldson, National Institutes of Health, USA

Professor Julie Donaldson, National Institutes of Health, USA
Julie Donaldson received her PhD from the University of Maryland in 1988 and did her postdoctoral research training at NIH before starting her own lab in 1995 at the National Heart, Lung and Blood Institute at the NIH. She is currently a Senior Investigator in the Cell Biology and Physiology Center. Her research program focuses on understanding the mechanism and physiological function of clathrin-independent endocytosis (CIE) and the subsequent routing and fate of endocytosed membrane proteins. CIE is the entry mechanism for many cell surface proteins including cell adhesion molecules, ion channels, nutrient transporters and microbial pathogens. Macropinocytosis is another form of CIE that provides a unique platform for following membrane dynamics.
| 13:30 - 14:00 |
Regulation of macropinocytosis in Ras-driven tumours

Dr Cosimo Commisso, NCI Cancer Center, Sanford Burnham Prebys Medical Discovery Institute, USA

Dr Cosimo Commisso, NCI Cancer Center, Sanford Burnham Prebys Medical Discovery Institute, USADr Cosimo Commisso earned his PhD in 2008 at the University of Toronto in the Department of Molecular Genetics and completed his Postdoctoral training at New York University Langone Medical Center in 2014. He is currently an Assistant Professor in the SBP Medical Discovery Institute NCI-Designated Cancer Center. Dr Commisso made a seminal contribution to the field of cancer research by determining that Ras-mutated tumors augment their glutamine supply through boosting macropinocytosis-dependent protein catabolism. Research in Dr Commisso’s lab is centered on identifying the underlying mechanisms that drive macropinocytosis in Ras-mutated tumors. Dr Commisso’s work has garnered several awards, including a Natural Sciences and Engineering Research Council of Canada award, an Ontario Scholar Award, two Canadian Institutes of Health Research awards, two AACR Pancreatic Cancer Action Network awards, a Cancer Centers Council Pedal the Cause Team Science Award and most recently a Career Development Award from the U.S. Department of Defense. |
|
|---|---|---|
| 14:00 - 14:15 | Discussion | |
| 14:15 - 14:45 |
Regulation and consequences of non-canonical autophagy resembling LC3-Associated Phagocytosis or LAP
Autophagy proteins were discovered by their regulation of macroautophagy but are now known to have parallel functions in related processes that also involve LC3 lipidation. Notably, LC3-Associated Phagocytosis or LAP is a parallel function of core autophagy proteins that involves lipidation of LC3 onto phagosomes and other endocytic membranes including macropinosomes. Dr Overholtzer's group has found that LAP is induced by changes to the osmotic environment of macropinosomes or endolysosomes. Here Dr Overholtzer will discuss new data uncovering regulation of LAP and the consequences of this process that differ from macroautophagy. The group finds that LAP induction leads to selective turnover of endolysosomal membrane and the ion channel TRPML1. 
Dr Michael Overholtzer, Memorial Sloan Kettering Cancer Center, USA

Dr Michael Overholtzer, Memorial Sloan Kettering Cancer Center, USAMichael Overholtzer is an Associate Member in the Cell Biology Program at Memorial Sloan Kettering Cancer Center. He received his bachelor’s degree from Ithaca College, and his PhD from Princeton University where he performed research on the p53 tumor suppressor with Arnold J Levine. In his postdoc with Joan S Brugge at Harvard Medical School he identified the Hippo pathway effector Yap as an oncogene in breast cancer, and discovered a cell death mechanism called entosis. In his independent laboratory he continues to study mechanisms of cell death, autophagy and lysosomes. |
|
| 14:45 - 15:00 | Discussion | |
| 15:00 - 15:30 | Tea | |
| 15:30 - 15:45 |
Strategies to inhibit the intracellular target Ras with potent antibody mimetic proteins
Antibodies, and antibody-like mimetic proteins, are known to be excellent therapeutic molecules for addressing extracellular human targets. However, less is known about their potential as inhibitors of intracellular targets, which make up two-thirds of the expressed human genome. Mutations in the intracellular target Ras are strong oncogenic drivers of many cancers but the target Ras is still not addressed by any current therapies. Dr Minter describes an antibody mimetic, DARPin K27, which blocks Ras by inhibiting nucleotide exchange of GDP to GTP. Intracellular expression of K27 significantly reduces the amount of active Ras, inhibits downstream signalling, in particular the levels of phosphorylated ERK and AKT, and slows the growth in soft agar of HCT116 cells. K27 is the most potent non-covalent inhibitor of nucleotide exchange reported, showing consistent effects across wild type and oncogenic mutant forms of Ras. In addition the group has isolated a second DARPin K55, which is selective for the active, GTP form of Ras and inhibits signalling by directly blocking interactions with downstream effector molecules such as Raf. The isolation and characterisation of these antibody-like molecules enable us to explore novel strategies to (i) deliver functional macromolecules into cells and (ii) understand the biological implications of Ras inhibition by different mechanisms. 
Dr Ralph Minter, Medimmune, UK

Dr Ralph Minter, Medimmune, UKFollowing a PhD in Immunology from the University of Durham in 1999, Ralph has been working in Antibody Discovery and Protein Engineering (ADPE) at MedImmune, developing biological therapies in the areas of oncology, autoimmune disease, asthma and infection. Several of these candidate drug molecules are now progressing through clinical trials (including the antibody Benlysta®, approved in 2011 for SLE). As Director, Fellow at MedImmune, Ralph actively pursues novel scientific and collaborative opportunities within the biologics field and has published 31 papers to date, covering diverse areas such as phenotypic drug discovery, antibody and protein engineering and novel target biology. His current focus is on the delivery of biological macromolecules into cells. |
|
| 15:45 - 16:00 |
Enhancing intracellular delivery of mRNA
Modified mRNAs offer a potentially transformative way of treating disease by triggering the body’s natural processes for protein production. A major challenge for therapeutic application is productive delivery of modified mRNA to cell cytoplasm for protein translation. The use of lipid nanoparticle (LNP) based vehicles to aid delivery of nucleic acids is well documented, however key challenges remain with lack of efficacy observed in some target tissue types associated with poor uptake and endosomal entrapment. Several mechanisms for LNP uptake have been proposed, including macropinocytosis however a detailed understanding remains elusive. This work aims to unravel the key mechanisms controlling cellular uptake and intracellular trafficking of LNPs and mRNA cargo across different cell lines. Dr Desai's group first screened uptake of LNPs and productive delivery of mRNA cargo across a range of tumour cell types. Cellular uptake and cytoplasmic delivery was monitored using fluorescently labelled LNPs delivering mRNAs encoding a reporter protein (eGFP). The group found delivery to be highly variable, with some tumour types completely refractile to LNP based delivery. The group then applied a phenotypic screening approach using selected libraries of small molecule compounds alongside RNA interference strategies with the objective to identify pathways that if perturbed would enhance LNP mediated delivery. This work has resulted in a greater understanding of the cellular mechanisms that control LNP based delivery and fate of mRNA cargo and will facilitate the design of next generation intracellular delivery technologies. 
Dr Arpan Desai, AstraZeneca, UK

Dr Arpan Desai, AstraZeneca, UKArpan completed his first degree in Biotechnology at the University of Edinburgh, following which he obtained a PhD from the AZ/University of Nottingham Doctoral Training Centre in Targeted Therapeutics, where he studied the uptake and intracellular trafficking pathways of polymeric nanoparticles using pH-based nanosensors. He joined AZ in 2013 where he has worked primarily in the field of nucleic acid delivery. He has played a leading role in AZs formulation and delivery development activities for therapeutic nucleic acids with a focus on understanding cellular delivery mechanisms of mRNA using non-viral delivery vehicles. He has been involved in a number AZs collaborations to develop nucleic acid based therapeutics including Moderna Therapeutics (mRNA), Ionis (ASOs) and Regulus (anti-miRs). More recently, he has relocated to Cambridge where he has been working within AZs newly formed Advanced Drug Delivery Group to build up capabilities around intracellular delivery of nucleic acids. |
|
| 16:00 - 16:15 | Discussion | |
| 16:15 - 17:00 | Overview and future directions |
