A new Journal of the Royal Society Interface study explores the role of modelling in investigating the human gut microbiota.
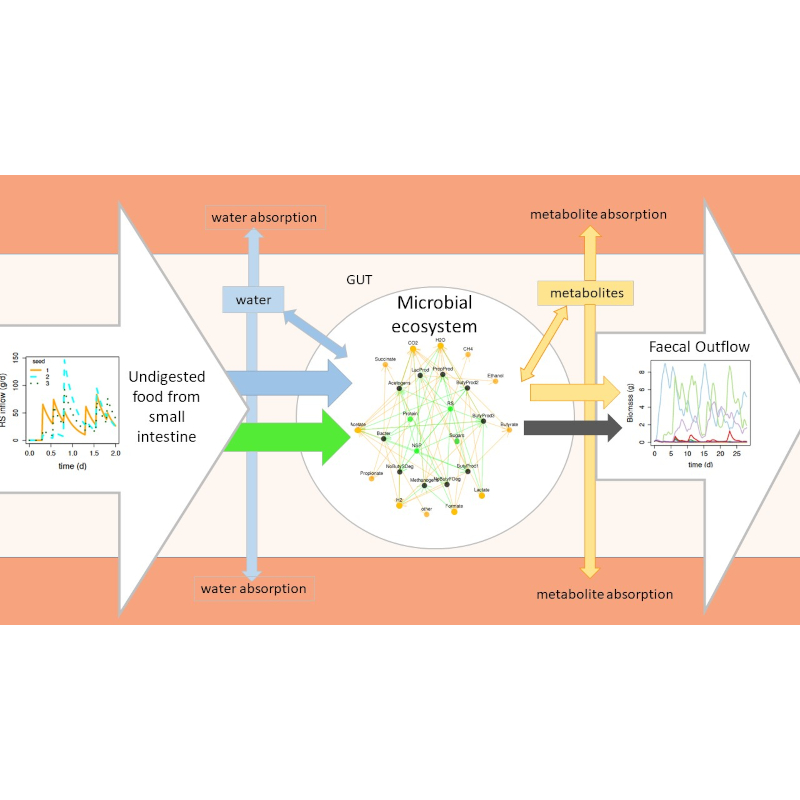
Mathematical modelling is a powerful tool that can be used to explore important research questions – often in tandem with experimental data. In a new Journal of the Royal Society Interface study, a team of researchers develop a model that simulates growth in the colon in response to different functional groups and diets.
Dr Helen Kettle from Biomathematics and Statistics Scotland explained to us about what the gut microbiota is, the clinical implications of the research and future research avenues.
1) Can you tell us about the background and importance of your work?
The human gut can contain about 10-100 trillion microbial cells meaning that they most likely outnumber our human cells! We are becoming increasingly aware that this microbial community plays a crucial role in human health via absorption of microbial growth products (metabolites) through the gut wall. Unfortunately, it is difficult to measure this community within the human body, so we usually rely on data from faecal samples to infer the composition of someone’s gut microbiota using nucleic acid sequencing. However, the causes and significance of variations in microbial composition, between individuals and with variations in diet, remain uncertain. To investigate these questions, we have developed a process-based mathematical model which simulates microbial growth in the colon. Several such models exist but our model has the most detailed microbial community with 10 different microbial functional groups which interact in response to meals (with varying composition) to give the metabolic profiles for different diets. We can also use the model to try “what if?” scenarios such as removing particular microbial groups and looking at the effect on the system. Furthermore, the open-source model code is available for other researchers to build on as more knowledge becomes available in the future.
2) How does your study support human volunteer research to enhance our understanding of the gut microbiota?
Our model represents a summary of what we currently know about our gut microbial ecosystem. By comparing this model with faecal data from human volunteers on fixed diets we can find out what we don’t yet understand and confirm what we do. When model simulations do not match the data we know that we have more to learn about the system and we can direct our research accordingly. Human volunteer research in combination with process-based modelling is crucial to try to unpick cause and effect when we don’t have direct access to the system within the human gut.
3) Can you comment on any public health or clinical implications of your research?
The ability to predict how dietary change affects the gut microbiota and gut metabolism is important not only in healthy individuals, but also in those suffering from digestive disorders or receiving medical treatment. Specifically, our observation and prediction that the proportion of butyrate increases as microbial fermentation of certain types of fibre increases, has important consequences in view of the multiple known impacts of butyrate on gut health.
4) What are the important questions that you think should be explored in the future?
There are a number of areas that could be important avenues to explore in helping us understand more about the gut microbiota. These include:
What are the health consequences of variations in gut microbiota composition between individuals?
Can we find better ways to continuously monitor metabolite concentrations, pH and microbial populations in different regions of the human gut?
How do different types of dietary fibre influence gut microbial growth and metabolism?
For more innovative research that uses modelling to address important biological questions, check out Journal of the Royal Society Interface.
Image credit: Schematic of the microbial ecosystem in the human colon. Helen Kettle.